Knowledge Center
July 12, 2023
What is Endoscope Reprocessing?

Endoscope processing is the process of cleaning and disinfecting reusable endoscopes before patient use. Due to the complex, intricate design, flexible and semi-rigid endoscopes should be handled properly to prevent damage, and device instructions for use (IFU) should be followed for proper reprocessing.
The process starts with point-of-use treatment, leak testing, manual cleaning, and recertification program. An automated endoscope reprocessor (AER) may perform cleaning to provide redundancy, followed by high-level disinfection (HLD) or liquid chemical sterilization (LCS). The process is then completed by drying and storing the endoscope prior to its next use. Transporting of the endoscope is performed at various stages of the workflow. Each process step is equally important to ensure the endoscope is safe for patient use.
Why is Endoscope Reprocessing Important?
Over 250 million endoscopies are performed each year globally
Minimally invasive procedures, like endoscopies, are growing by leaps and bounds every year. Over 250 million endoscopies are performed globally each year, with this number continuing to grow year over year1. With the increase of these procedures, regulations, standards, and guidelines have been established. To reduce the risk of infections using these reusable devices and to ensure the highest level of patient care and safety.
Endoscope Processing Steps
What are the 6 basic steps required for reprocessing a flexible endoscope?
- Point-of-Use Treatment
- Leak Testing
- Manual Cleaning and Verification
- HLD or LCS
- Endoscope Drying and Storage
- Transporting of Endoscopes
Endoscope Point-of-Use Treatment
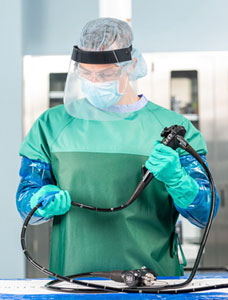
Point-of-Use Treatment occurs in the procedure room immediately after the procedure. If soils are given the time to dry, they become much more challenging to remove and can decrease the effectiveness of disinfection or sterilization.
Point-of-use treatment removes organic material (e.g., blood, body fluids) and decreases bioburden, making it much more likely that subsequent reprocessing steps will be successful2. Point-of-use treatment can be performed using a non-linting cloth, wipe, non-abrasive sponge, and a compatible cleaning agent. For example, the Revital-Ox™ Bedside Complete Pre-Cleaning Kit comes with a sponge and pre-diluted detergent, making both ready to use at the bedside immediately following a procedure.
Endoscope Leak Testing
Following the endoscope's arrival in the reprocessing area, leak testing should be performed as soon as possible. Leak testing allows the technician to detect potential damage to an endoscope's external surfaces and internal channels. There are multiple types of leak testing, including manual and automated, and reprocessing staff must follow the manufacturer's IFU to ensure the endoscope maintains its watertight integrity.
- A dry leak test pressurizes the endoscope, and a failed leak test is detected by sensors identifying a decrease in pressurization.
- A pressurized endoscope is submerged in clean water for a wet leak test to check for bubbles. If bubbles appear, this signals a leak from the endoscope.
The endoscope should be sent for repair, following facility policy and procedures if damage is detected during a leak test.
Endoscope Manual Cleaning and Verification
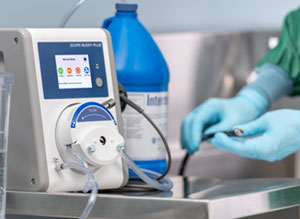
Once the leak test has been performed, employees should follow the endoscope manufacturer’s validated cleaning process. Manual cleaning is the most critical step when reprocessing an endoscope. As the saying goes: if it is not clean, it cannot be high-level disinfected or sterilized. Manual cleaning includes brushing and flushing the endoscope’s channels and ports. The process is performed at the sink with a compatible detergent, such as Revital-Ox™ Enzymatic Detergents.
It is also essential to perform the manual cleaning step within the timeframe specified by the endoscope manufacturer. If reprocessing is delayed, the endoscope should be cleaned per the manufacturer’s written IFU for delayed processing, which often has additional steps and requires more technician time.
During the endoscope cleaning procedure, healthcare personnel should always follow the device and detergent manufacturer’s IFU for temperature, concentration, and water quality. Once the cleaning solution has been prepared, all removable parts and accessories of the endoscope should be detached and immersed. The process is completed by cleaning the exterior, brushing all appropriate channels until debris is not present, flushing with enzymatic cleaning solution, rinsing the endoscope, and finally, an air purge.
Manual cleaning is the most critical step of reprocessing, and when point-of-use treatment and manual cleaning steps are correctly followed, the number of pathogens detected on endoscopes can be decreased by 99.9%3.
After manual cleaning, staff should visually inspect the endoscope to identify damage or debris. It is also recommended the endoscope be inspected using a magnifier and/or borescope to inspect where the unaided eye cannot see, including the inside channels of the endoscope. If any debris or damage is found, the endoscope should be recleaned or repaired.
Qualitative Protein Detection tests can be completed to assist with inspection. Some tests allow you to test medical device surfaces, including crevices and ports, for the presence of protein. Whether it's testing the exterior or lumen of a device, you can confirm if residual protein soils are present after the cleaning process, prior to high-level disinfection or sterilization.
Learn more about Endoscope Manual Cleaning
High-Level Disinfection or Liquid Chemical Sterilization
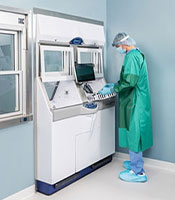
The next step in the endoscope reprocessing phase is HLD or LCS. Per ST91, personnel should refer to the medical device manufacturer’s written IFU and the device’s intended use to determine the method for a specific device. “Semi-critical devices come in contact with mucus membranes or non-intact skin. These items should be thoroughly cleaned and then sterilized. If sterilization is not possible, high-level disinfection is the minimum advised processing method.”4
While high-level disinfection can be performed by manually soaking the device, it is recommended that the process be undertaken in an AER. An example of an AER is the ADVANTAGE PLUS™ Pass-Thru Automated Endoscope Reprocessor. This AER also supports a pass-thru workflow design to help minimize the risk of cross-contamination.
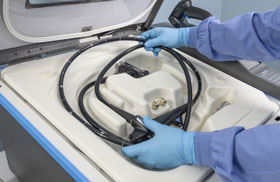
LCS can be performed using a processor such as SYSTEM 1™ endo Liquid Chemical Sterilant Processing System (LCSPS). The processor is intended for cleaned, immersible, and reusable semi-critical heat-sensitive medical devices and their accessories. During the cycle, the device and channels are exposed to several processing phases, including LCS. At the end of a successful cycle, the endoscope is free of all viable microorganisms, including bacterial endospores.
Endoscope Drying and Storage

Once HLD and LCS have been performed, it is time for drying and storage. Drying is a crucial step in endoscope reprocessing to ensure no moisture is left on the endoscope. Moisture can allow microorganisms and biofilm to survive and multiply. Even when reprocessing steps are performed meticulously, a few microorganisms may survive HLD. Those few microorganisms can multiply to over a million colony-forming units in a few hours if moisture remains in the endoscope channels or on its surface6.
How do you dry and store endoscopes? Device IFUs must be followed through all process steps, including requirements and steps for drying prior to placement in the endoscope drying cabinet.
For storage, Per ST91:2021: “endoscope storage requirements include that endoscopes be suspended vertically or horizontally in a cabinet designed for storage to allow air circulation and provide channel purging in accordance with the endoscope manufacturer’s written IFU.”
Transporting Reprocessed Endoscopes

Finally, once the endoscope has been dried and/or stored, it must be cautiously handled when transported to the procedure room. If not, endoscopes risk becoming re-contaminated and will need to go through each step of the reprocessing phase.
Hand hygiene should be performed before applying new gloves and handling the endoscope. During transport, the endoscope should be identified as clean and protected from contamination and damage. Unless transported through a controlled, connected corridor, the endoscope should be loosely coiled and placed in a clean, covered, solid endoscope transport container large enough to accommodate a single endoscope without the need to over-coil the insertion or light guide tubes7. The endoscope is then ready for patient use.
Proper endoscope reprocessing is imperative for the best patient outcomes and the safety of your staff, with each of these six essential steps as critical as the next. With the increasing number of flexible endoscopies being performed, a successful and efficient process is paramount, and STERIS offers a solution for every step of the process.
Endoscope Reprocessing Guidelines & Standards
Endoscope reprocessing standards and guidelines are created by organizations like the Association for the Advancement of Medical Instrumentation® (AAMI), the Association of periOperative Registered Nurses (AORN), and The Society of Gastroenterology Nurses and Associates (SGNA). These standards, guide healthcare personnel involved in the reprocessing of endoscopes. Staff must be educated, trained, and complete competency verification assessments based on the endoscope manufacturer’s IFUs, the reprocessing equipment, and the chemicals used.
Six essential steps must be followed during reprocessing to confirm that an endoscope is safe for patient use. The proper knowledge and tools are crucial for the best patient outcomes. ANSI/AAMI ST91:2021 – The comprehensive guide to flexible and semi-rigid endoscope processing in healthcare facilities was published to provide a comprehensive guide to achieve the best practice for each stage of processing flexible endoscopes:
“To achieve best practice for each stage of processing flexible endoscopes as well as competency and education of staff, meticulous attention to all steps in the processing of flexible endoscopes and accessories is critical to ensure that they are rendered safe for subsequent patient use.”
Explore STERIS Products that Support Endoscope Reprocessing
Article References
1 A Surprising Over 250 million Endoscopies Are Performed Each Year Globally, September 2022 - https://www.strategicmarketresearch.com/blogs/endoscopies-performed-each-year
2, 6 Standards of Infection Prevention in Reprocessing Flexible Gastrointestinal Endoscopes, SGNA 2018
3 GI Endoscope Reprocessing: A Comparative Review of Organizational Guidelines and Guide for Endoscopy Units and Regulatory Agencies, GIE 2022 https://www.giejournal.org/article/S0016-5107(21)01655-2/fulltext
4, 5, 7 ANSI/AAMI ST91:2021 Flexible and Semi-Rigid Endoscope Processing in Healthcare Facilities, American National Standard 2022