Knowledge Center
February 13, 2023
What are Retained Surgical Items?
Retained surgical items (RSIs), sometimes referred to as retained foreign bodies or retained foreign objects (RFOs), are any instruments, tools, devices, or surgical gauze unintentionally left in the body cavity at the completion of a surgery or other procedure, including vaginal sponges remaining after procedures or deliveries. This article will highlight the inherent risks retained surgical items pose to patient safety and ramifications they have on healthcare organizations, and explain how to help prevent an unintended retained surgical item from occurring.
Unintentionally retained surgical items remain a high risk to patient safety despite thorough processes and tedious effort by operating room (OR) staff to ensure that items used during a surgical procedure are accounted for prior to skin closure. Typical processes for tracking RSIs are centered around manual counts, which can be inaccurate, and items such as missed sponges left in a patient can lead to additional surgeries, pain, and suffering.
The number of annual incidences of RSIs in the United States varies due to many factors, including underreporting or inconsistent requirements, lack of a concrete definition of RSIs, varying use of RSI prevention methods, and differences in procedural settings (i.e., hospitals, ambulatory surgery centers). One study of 5 non-specialty acute care hospitals found the incidence varied from 1 in every 8,801 to 1 in every 18,760 inpatient operations – corresponding to 1 case or more each year for a typical large hospital.1
Help Prevent Retained Surgical Items with the Surgical Counting and Detection System
Different Types of Retained Surgical Items
The different types of retained surgical items vary, and generally refer to any items left in a patient after a surgery or procedure that were intended to be removed.2 Some examples include forceps, tips for foley catheter insertion, drill tips and other device fragments, and surgical gauze. If a surgical sponge, instrument or tool is retained after the procedure, negative consequences may result for both the patient and the health care facility. The unintended retention of surgical items can be a root cause for hospital readmission, the need for X-rays, reoperation and, rarely, death.3
Among the various types, the most common occurrence of RSIs are “gossypiboma” which is a surgical sponge, or a laparotomy pad left involuntarily in the body after a procedure. Retained surgical sponges account for 48% to 69% of RSIs, per a 2019 report from the ECRI Institute.4
Clinical Presentations and Technical Investigation

Cotton and synthetic medical sponges can present more detectable tissue reactions than metal fragments. The patient’s tissue may respond to this foreign material with an aseptic fibrous response (e.g., adhesions, encapsulation, or granulomas) which develops relatively slowly, or, if the sponge is in a nonsterile location like the gastrointestinal tract, with an early exudative inflammatory response that may result in an abscess or fistula.
Some early symptomatic indicators of RSIs are unexplained pain, features of generalized sepsis, and abscess formation. Symptomatic response that is delayed might include nonhealing wounds and signs of intestinal obstruction or internal fistulization, among others. Of course, patients may also be asymptomatic and in such cases detection of RSIs is incidental.
The abdomen and pelvis are the most common locations where RSIs are found, but there have also been reports of retained surgical sponges in the thorax, spine, head and neck, brain, and extremities. Sponges also may be retained in natural orifices, such as the vagina or nose, in minimally invasive surgeries and after vaginal births.2
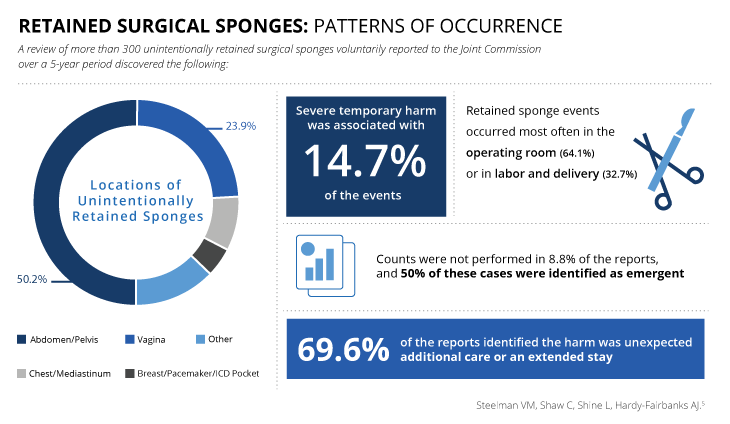
A review of more than 300 unintentionally retained surgical sponges voluntarily reported to the Joint Commission over a 5-year period discovered the following:5 Sponges were most frequently retained in the abdomen or pelvis (50.2%) and in the vagina (23.9%). Retained sponge events occurred most often in the operating room (64.1%) or in labor and delivery (32.7%). Counts were not performed in 8.8% of the reports, and 50% of these cases were identified as emergent. In 69.6% of the reports, the harm identified was unexpected additional care or an extended stay. Severe temporary harm was associated with 14.7% of the events.
What are the Causes of Unintentionally Retained Surgical Items?
Facilities are expected to conduct a root-cause analysis in the event of an unintentionally retained surgical item. Frequently, events of RSIs fall under categories of human interactions, leadership and communication. According to event data on RSIs reported to the Joint Commission from 2005 to 2012, the most common root causes of RSIs were:
- Absence of policies and procedures, or failure to comply with existing policies and procedures
- Problems with hierarchy and intimidation
- Failure in communication with physicians
- Failure of staff to communicate relevant patient information; and,
- Inadequate or incomplete education of staff
In addition, 88% of retained sponges occur with what staff believe is a correct count.1 If staff wrongly concludes that all sponges have been accounted for, the surgical team may not learn of the discrepancy unless the patient returns with a complaint of pain or discomfort.
Risk of Occurrence of Retained Surgical Items
The opportunity for RSIs exists even when a procedure involves very small incisions2 and is not exclusive to higher risk procedures. However, there are common risk factors for retained surgical items to be aware of, which call for heightened awareness of the potential for RSIs:
- Patient with a high body mass index (BMI)
- An emergent or urgent procedure (RSIs are 9 times more likely with emergency operations)
- Intra-abdominal surgery
- Unanticipated/unexpected change during the procedure (e.g., a change in approach/incision, type of procedure, or complications developed during the procedure)
- The involvement of multiple surgical teams and staff turnovers during the procedure; and
- Execution of multiple surgical procedures or long procedure duration
Impacts on the Healthcare Facility
On top of the risk to patient safety, incidences of RSIs can also carry significant financial and public relations impacts on the healthcare facility, including increased costs of care, threat of litigation, and harm to the institution’s public image.
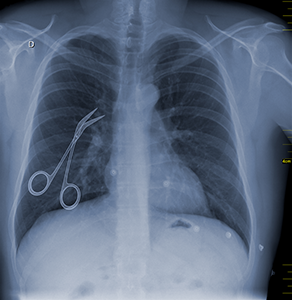
Contributing factors to the increased cost of an RSI include additional OR time to resolve miscounts (averaging 13 minutes per event), as well as time spent completing a root-cause analysis for the event, reporting requirements, and the identification and implementation of new solutions. Studies have estimated that the average total cost of care related to an RSI is approximately $166,000 to $200,000 per incident, including the cost of the hospital’s legal defense, indemnity payments, and surgical costs not reimbursed by the Centers for Medicare & Medicaid Services (CMS).
RSI cases are avoidable and, because an object unintentionally left in a body cavity where it does not belong is frequently injurious, many lead to malpractice claims. Members of the entire surgical team, as well as the health care facility, can be held liable in RSI litigation.2 Whether a settlement is reached prior to litigation or not, it is still an important area of risk mitigation for a healthcare facility.
Additionally, the retention of sponges can attract widespread, critical press coverage. The institution's reputation may be negatively affected by the publicity surrounding mandatory public reporting of such adverse patient events.6
In many instances, the rarity of adverse events like RSIs tends to reinforce the surgical team’s belief that they will never experience them, which can lead to a misplaced confidence that existing safety systems are adequate. This complacency itself is a threat to safety, which further increases the risk of RSIs.
How to Prevent Retained Surgical Items
Healthcare organizations are responsible for employing standardized, verifiable practices to account for all surgical items used during a procedure. Steps that can be taken include systematic manual counting of material during the procedure, standardized count reconciliation procedures, methodical wound exploration, radiological confirmation, and the use of assistive technologies, such as radio frequency identification (RFID) tracking devices.
Beyond the implementation of practices, preventing RSIs requires a comprehensive approach that involves the circulator, scrub person, surgeons and surgical first assistants, and anesthesia professionals. Good communication among the staff in the operating environment, and transparent reporting are critical to reducing incidences of RSIs.
Adjunct Technology and Retained Surgical Items
Despite modern advances in surgical technology, teams often rely solely on manual counting protocols – in which the surgical team verifies all sponges are accounted for before concluding the procedure – as the main safeguards to prevent RSIs. Although manual counts are an accepted standard practice, they are also prone to error. Studies have shown that current practices for counting medical sponges have a 10% to 15% error rate.7
Many incorrect counts are false alarms, and reconciling an incorrect count prolongs the procedure. If a discrepancy with the count is identified, the use of X-rays and involvement of radiology staff are required to locate the missing item, adding to patient risk.
Recently, the Association of periOperative Registered Nurses (AORN) published updated Guidelines for Prevention of Unintentionally Retained Surgical Items recommending the use of adjunct technology during manual counting, to prevent RSIs.2 Several new technologies have emerged that can help OR staff virtually eliminate RSIs.3 These technologies are designed to enhance counting protocols, rather than be a substitute.
Help Avoid Missed Counts!
Download our Top 8 Considerations When Evaluating Technology for Preventing Retained Surgical Items.
- A count discrepancy occurs in 1 out of every 8 cases
- A miscount occurs in 1 out of every 65 procedures
- Retained Surgical Items occur 39 times per week in the U.S.
Think your operating room can’t be among them?
Get our checklist of the Top 8 things to consider when evaluating adjunct technology to assist in your surgical counts, and follow AORN guidelines recommending the use of technology during manual counting.
*By filling out this form, you are opting in to receive
emails from STERIS.
Benefits of Using RFID Tracking for Surgical Items
The unique advantage of RFID systems over other adjunct technologies is their ability to count, locate, and identify surgical items using unique serial numbers, whereas barcode-based systems simply count the items and RF-based systems can only locate them. An RFID system is typically comprised of specialized tags that enable unique identification of individual surgical items, a movable workstation, a handheld reader, a locator wand that can detect tagged items, and management software that connects with other business intelligence applications.
This allows for integration with OR and sterile processing workflows and real-time feedback for improved department efficiency. The primary benefit of using RFID technology for tracking RSIs is higher accuracy of detection, eliminating false positives and false negatives. This technology can help save time and human effort used during counting protocols, and has the potential to substantially improve patient safety by reducing RSI errors.8
ORLocate Surgical Counting and Detection System
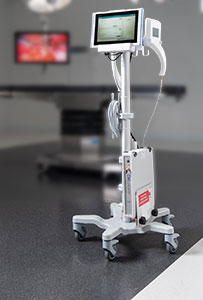
The ORLocate Surgical Counting and Detection System is one of these such technologies available today. Within seconds, ORLocate has the ability to count multiple surgical items before, during, and after procedure. It also enables the user to detect items within the patient cavity as well as other locations in the OR (e.g., waste bins, kick buckets) protecting against missed counts and reducing near misses.
Retained surgical items can be further prevented with the use of assistive technology and stringent counting practices. New technologies that supplement manual counts have been found to be effective when used correctly, and healthcare professionals are encouraged to adopt these innovations.
Contributors

Lena Fogle BSN, RN, CNOR
Senior Director Global Clinical Solutions, STERIS Healthcare
![]()
Lena is a seasoned healthcare leader with extensive experience leading complex perioperative environments as well as new program development, continuous process improvement, clinical outcomes, operational excellence, and stakeholder experience.
Related Resources
Article References:
1 Gawande AA, Studdert DM, Orav EJ, Brennan TA, Zinner MJ. Risk factors for retained instruments and sponges after surgery. N Engl J Med. 2003;348(3):229-235. https://pubmed.ncbi.nlm.nih.gov/12529464
2 Association of periOperative Registered Nurses. Guideline for Prevention of Retained Surgical Items. In: Guidelines for Perioperative Practice. https://aornguidelines.org/guidelines?bookid=2260. Denver, CO: AORN; Revised: December 2021.
3 Sadler D. New technologies help prevent RSIs. https://ortoday.com/new-technologies-help-prevent-rsis/. Published February 1, 2012.
4 ECRI Institute. 2019 Top 10 Health Technology Hazards: Executive Brief. https://www.ecri.org/Resources/Whitepapers_and_reports/Haz_19.pdf. Published 2018.
5 Steelman VM, Shaw C, Shine L, Hardy-Fairbanks AJ. Retained surgical sponges: a descriptive study of 319 occurrences and contributing factors from 2012 to 2017. https://pssjournal.biomedcentral.com/articles/10.1186/s13037-018-0166-0. Patient Saf Surg. 2018;12:20.
6 Cima RJ, Kollengode A, Clark J, et al. Using a data-matrix-coded sponge counting system across a surgical practice: Impact after 18 months. https://www.jointcommissionjournal.com/article/S1553-7250(11)37007-9/fulltext. Joint Commission Journal on Quality and Patient Safety. 2011;37(2):51-58, AP51-AP53.
7 Joint Commission. Preventing unintended retained foreign objects. https://www.jointcommission.org/-/media/tjc/documents/resources/patient-safety-topics/sentinel-event/sea_51_urfos_10_17_13_final.pdf. Published October 17, 2013.
8 Schnock KO, Biggs B, Fladger A, Bates DW, Rozenblum R. Evaluating the impact of radio frequency identification retained surgical instruments tracking on patient safety. https://pubmed.ncbi.nlm.nih.gov/28230583. Literature review. J Patient Saf. 2017.



