Knowledge Center
June 16, 2022
Low Temperature Sterilization: Throughput and Material Compatibility
In a head-to-head comparison of V-PRO vs. STERRAD™, it's clear that V-PRO beats STERRAD™ with a one-two punch in both throughput and material compatibility. This winning combination allows your Sterile Processing Department (SPD) to process most instruments faster to optimize your workflow and productivity.
Download the V-PRO vs. STERRAD™ Infographic
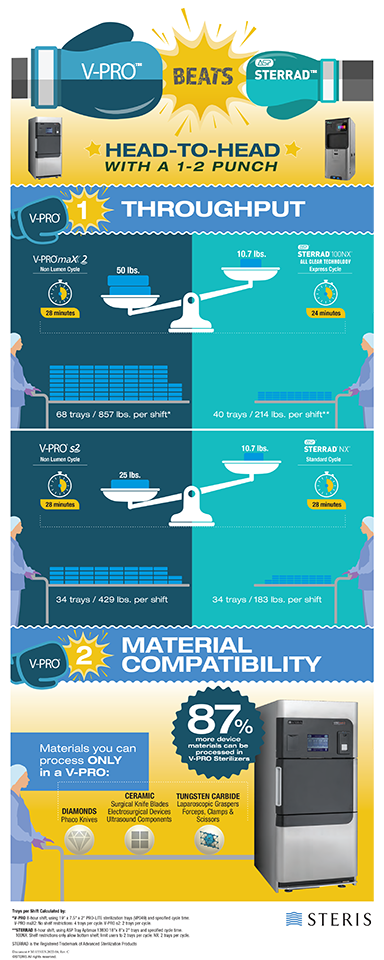
Sterilizer Throughput
Throughput is a measurement of the productivity of a sterilizer, factored by cycle time and the volume of surgical instruments the system can process in an 8-hour shift. To determine which low temperature sterilizer is ideal for your facility’s required throughput, key considerations should include your inventory of devices and your turnover requirements (how fast do you need the devices). Use our infographic above to help understand how changes in weights, or cycle time, can affect your overall throughput.
Material Compatibility
Material compatibility is important, because the more materials that you can process in your low temperature sterilizer the more devices that may be validated for that system. A device’s Instructions for Use (IFU) will indicate compatible sterilization methods, such as vaporized hydrogen peroxide (VHP) sterilization, and materials used in that medical device determine compatibility.
STERIS's V-PRO™ Low Temperature Sterilization Systems are the only* low temperature sterilization systems that are validated to sterilize up to 43 different materials, including:
- Diamonds (Device Example: Phaco Knives)
- Gold (Found in Cables and Sensors)
- Ceramic (Surgical Knife Blades, Electrosurgical Devices, Ultrasound Components)
- Tungsten Carbide (Laparoscopic Graspers, Forceps, Clamps & Scissors)
Confirm your medical devices and accessories are compatible with V-PRO™ Low Temperature Sterilization Systems with STERIS’s Device Compatibility Matrix and follow all device IFUs for processing.
Related Resources
*When compared to STERRAD™ Systems and Sterizone® VP4 Sterilizers, as of July 22, 2021.