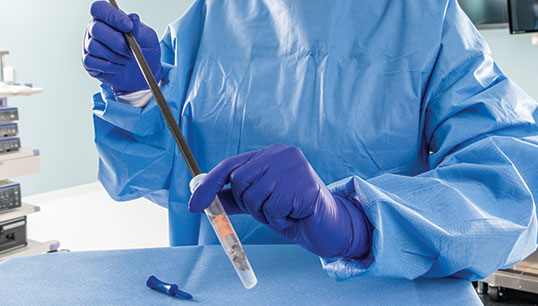
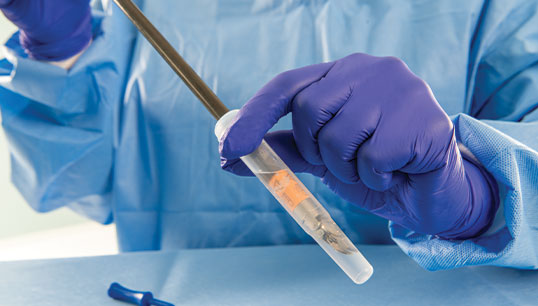
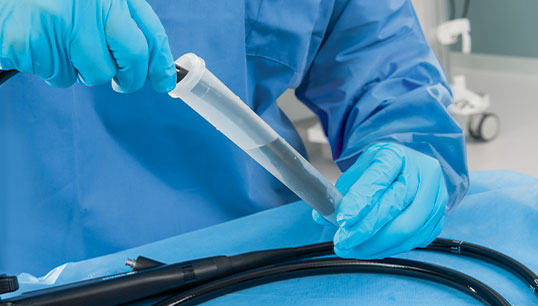
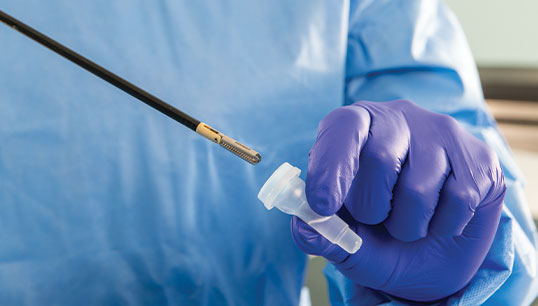




PRE-KLENZ™ Soak Shield
- Initiates the pretreatment process of robotic, surgical, and endoscopic instruments at the bedside
- Provides device tip protection while simultaneously soaking and cleaning during transport
- Ready-to-use, single-use application allows for easy, quick application
Product Overview
PRE-KLENZ Soak Shield helps initiate the pre-cleaning process of surgical, endoscopic, and robotic instruments at the bedside immediately after use and continues through transport. The device tip is submerged in enzymatic detergent from the bedside through transport to loosen soils, making reprocessing easier. PRE‑KLENZ Soak Shield is available in small, medium, and large sizes depending on the device's needs.
| Large | Medium | Small |
|---|---|---|
 |
 |
 |
| For use with larger flexible devices such as duodenoscopes, colonoscopes, gastroscopes, sigmoidoscopes, etc. | For use with rigid, semi-rigid, and smaller flexible devices such as robotic instruments, bronchoscopes, cystoscopes, ureteroscopes, forceps, arthroscopes, etc. | For use with rigid devices such as ENT devices. |
PRE-KLENZ SOAK SHIELD KIT
Everything you need for point of use treatment is in a complete kit.
- Maintains moisture in a humidity bag for up to 72 hours to help support an easier manual cleaning process
- Keeps instrument sets together during transport while protecting the device tip
Kit Includes:
- 5 medium PRE-KLENZ Soak Shield vials protect device tip and initiate pre-cleaning
- 100ml bottle of enzymatic solution to flush ports
- Large humidity bag keeps instruments moist during transport
- A puncture tool to break the seal of the Soak Shield membrane

How the PRE-KLENZ Soak Shield Works
With the device's tip encased, enzymatic detergent proactively moistens and loosens soils. Instrument pre-cleaning is active from the bedside through transport.
Why PRE-KLENZ Soak Shield?
- Promotes soil removal – Pre-moistens soils at the tip of the device and all exposed surfaces of surgical, endoscopic, and robotic instruments
- Immediate and continuous pre-cleaning – Powerful performance of STERIS enzymatic detergent
- Device tip protection – The design protects small, medium, and large surgical and endoscopic devices during transport
- Time-efficient – Transport now becomes valuable pre-cleaning time
California Ingredient Disclosure
Shop Now
Browse a complete Item Number list and easily reorder PRE-KLENZ™ Soak Shield products on Shop STERIS.
| Product Number | Description | |
|---|---|---|
| 2D66J3 | PRE-KLENZ SOAK SHIELD (SM 1 ML - 250 PER CASE) | |
| 2D66J4 | PRE-KLENZ SOAK SHIELD (MED 6 ML - 250 PER CASE) | |
| 2D66J6 | PRE-KLENZ SOAK SHIELD (LARG 35 ML - 4-CARTONS OF 25 100 PER CASE) | |
| 2D66Q5 | PRE-KLENZ SOAK SHIELD KIT (12 KITS PER CASE) |
|
SDS
|
|
|---|---|
| Document # | Document Title |
| PRE-KLENZ SOAK SHIELD SDS - US | |
| PRE-KLENZ SOAK SHIELD - AUSTRALIAN | |
| PRE-KLENZ SOAK SHIELD-SDS-CALIFORNIA | |
| PRE-KLENZ SOAK SHIELD-SDS-CANADA | |
| PRE-KLENZ SOAK SHIELD-SDS-CANADA | |
| PRE-KLENZ SOAK SHIELD-SDS-SWITZERLAND | |
| PRE-KLENZ SOAK SHIELD-SDS-GERMANY | |
| PRE-KLENZ SOAK SHIELD-SDS-DENMARK | |
| PRE-KLENZ SOAK SHIELD-SDS-FRANCE | |
| PRE-KLENZ SOAK SHIELD-SDS-FRANCE | |
| PRE-KLENZ SOAK SHIELD-SDS-JAPANESE | |
| PRE-KLENZ SOAK SHIELD-SDS-NETHERLANDS | |
| PRE-KLENZ SOAK SHIELD-SDS-NORWAY | |
| PRE-KLENZ SOAK SHIELD-SDS-SWEDEN | |
| PRE-KLENZ SOAK SHIELD-SDS-UNITED KINGDOM | |
| PRE-KLENZ SOAK SHIELD-SDS-AUSTRALIAN | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-UNITED STATES | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-AUSTRALIA | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-GERMAN | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-DANISH | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-FRENCH | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-JAPANESE | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-DUTCH | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-NORWEGIAN | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-SWEDISH | |
| PRE-KLENZ SOAK SHIELD KIT-SDS-UNITED KINGDOM | |
|
INSTRUCTIONS FOR USE
|
|
|---|---|
| Document # | Document Title |
| PRE-KLENZ SOAK SHIELD IFU | |
| PRE-KLENZ SOAK SHIELD KIT IFU | |
|
LABELS
|
|
|---|---|
| Document # | Document Title |
| PRE-KLENZ SOAK SHIELD SMALL 1MLX250 | |
| PRE-KLENZ SOAK SHIELD MEDIUM 6MLX250 | |
| PRE-KLENZ SOAK SHIELD LARGE 35MLX25 | |
| PRE-KLENZ SOAK SHIELD KIT - LABEL | |
|
LABORATORY REPORTS
|
|
|---|---|
| Document # | Document Title |
| PRE-KLENZ SOAK SHIELD SUBSTRATE COMPATIBILITY | |
|
BROCHURE
|
|
|---|---|
| Document # | Document Title |
| GI REPROCESSING PORTFOLIO BROCHURE | |
| PRE-KLENZ FAMILY BROCHURE | |
| PRE-KLENZ POINT OF USE PROCESSING GEL (AUTOMATED TRIGGER) (FRENCH) | |
| PRE-KLENZ SOAK SHIELD BROCHURE | |
| PRE-KLENZ SOAK SHIELD BROCHURE | |
| PRE-KLENZ SOAK SHIELD BROCHURE | |
|
WALL CHARTS
|
|
|---|---|
| Document # | Document Title |
| SOAK SHIELD KIT WALL CHART | |




