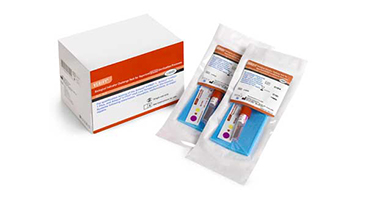
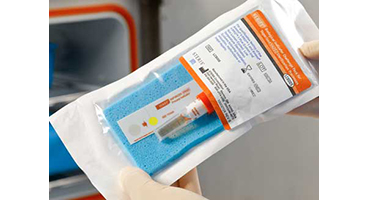
Check out the new:
Celerity™ 20 HP Challenge Pack
With fast 20-minute BI final read, you can perform qualification of a V-PRO Sterilizer in less than four hours
VERIFY® V24 Challenge Packs provide the confidence you need that your V-PRO Low Temperature Sterilization Systems are functioning properly after installation or major repairs. With the VERIFY V24 Challenge pack, only three consecutive cycles (most challenging) need to be run in a V-PRO Sterilizer to qualify the sterilizer for use.
How VERIFY V24 Challenge Packs Work
Each challenge pack contains a process indicator strip, VERIFY V24 Self Contained Biological Indicator and blue polyurethane foam sheet.
The indicator strip is a Type 1 Process Indicator as defined in ANSI/AAMI/ISO 11140-1 and provides immediate proof of processing. A vivid color change from magenta to yellow indicates that exposure to a hydrogen peroxide sterilization cycle has occurred.
Each self-contained biological indicator is completely self-contained, providing 106 colony forming units (CFU) of Geobacillus stearothermophilus and an ampoule of specifically formulated growth medium with a pH indicator. It is designed to be sealed after processing, reducing the risk of contamination (and consequent false positives) and evaporation of growth medium.
Following incubation, a vivid color change of the growth media from orange to yellow and/or turbidity gives distinct evidence of microbial growth. If no microbial growth occurs, the media remains orange and clear (no turbidity).
The challenge pack is placed on the bottom shelf within the empty chamber of the V-PRO Sterilizer. The sterilizer must expose the pack to a sufficient quantity of hydrogen peroxide to overcome the absorption of hydrogen peroxide from the polyurethane foam sheet and kill the bacterial spore contained within the self-contained biological indicator.
The challenge to sterilization provided by the pack is equivalent to or greater that that achieved by the worst case sterilization load.
Why use the VERIFY V24 Challenge Pack?
- Easy to Read – Orange to yellow color change indicates growth
- Tougher challenge than validated sterilization loads – Confidence in the V-PRO Sterilizer's performance
VERIFY V24 Challenge Pack's validated V-PRO sterilization cycles
| STERILIZER |
STERILIZER QUALIFICATION CYCLE |
CYCLE TIME |
|
V-PRO® s2 Low Temperature Sterilizer
|
Fast Cycle |
19 minutes |
| V-PRO® maX 2 Low Temperature Sterilizer |
Fast Non Lumen Cycle |
16 minutes |
|
V-PRO® maX Low Temperature Sterilizer
V-PRO® 60 Low Temperature Sterilizer
V-PRO® 1 Plus Low Temperature Sterilizer
|
Non Lumen Cycle |
28 minutes |
|
V-PRO® 1 Low Temperature Sterilizer
|
Lumen Cycle
|
55 minutes |
Browse a complete Item Number list and easily reorder VERIFY® V24 Challenge Pack products on Shop STERIS.
| Product Number |
Description |
|
| LCB005 |
VERIFY V24 CHALLENGE PACK (5 PKS & 5 CONTROLS/BX) |
|
An incubator and the VERIFY Self-Contained Biological Indicator HP Activator Set are required for proper use of the VERIFY Biological Indicator Challenge Pack for VH2O2 Sterilization Processes.
| Product Number |
Description |
Quantity |
|
|
LCB004
|
VERIFY™ Self-Contained Biological Indicator HP Activator Set
|
Each
|
|
|
S3080
|
VERIFY™ 28 Well Dual Incubator (55-60°C and 35-39°C)
|
Each
|
|
|
S3082
|
VERIFY™ 28 Well Incubator (55-60°C)
|
Each
|
|
|
S3271
|
VERIFY™ 6 Well Incubator (55-60°C)
|
Each
|
|
|
TECH DATA SHEET
|
| Document # |
Document Title |
|
|
VERIFY DUAL TEMPERATURE 28-WELL INCUBATOR (220 VAC) FOR VERIFY SELF-CONTAINED BIOLOGICAL INDICATORS (S3081)
|
|
|
VERIFY V24 CHALLENGE PACK
|
|
SAMPLE PROCEDURES
|
| Document # |
Document Title |
|
|
SAMPLE PROCEDURE-MICROBIAL VERIFICATION FOLLOWING INSTALLATION OR MAJOR REPAIR OF V-PRO 1, 1 PLUS, & MAX & V-PRO 60-FRENCH CANADIAN
|
|
|
SAMPLE PROCEDURE: MICROBIAL VERIFICATION FOLLOWING INSTALLATION OR MAJOR REPAIR OF V-PRO 1, 1 PLUS, & MAX &V-PRO 60-CANADA
|
|
SDS
|
| Document # |
Document Title |
|
|
VERIFY V24 AND V24I SELF-CONTAINED BIOLOGICAL INDICATOR AND CHALLENGE PACKS
|
|
|
VERIFY V24 AND V24I SELF-CONTAINED BIOLOGICAL INDICATOR AND CHALLENGE PACKS - CANADIAN
|